Protein Domains
This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What are protein domains?
|
Protein domains are regions of a protein sequence that have a specific structure and function [1]. These protein domains have distinct folding that allows them to interact with other proteins in a different way than other parts of that same gene. While homologous regions of a protein sequence suggest similar function, protein domains suggest similar interactions with other proteins [2].
|
What protein domains of VHL are present in the humans?
Low complexity region: are regions that have low diversity in their amino acid sequence. These regions may play a specific role in protein function [3].
VHL beta domain: bind Cul2 and is involved in regulation of vascular endothelial growth factor mRNA. It also forms a ternary complex with the elonginB and elonginC proteins, which regulates levels of HIFs [4].
VHL box domain: short alpha-helix domain that interacts with Cul2-Rbx1 to determine its binding specificity [5].
VHL beta domain: bind Cul2 and is involved in regulation of vascular endothelial growth factor mRNA. It also forms a ternary complex with the elonginB and elonginC proteins, which regulates levels of HIFs [4].
VHL box domain: short alpha-helix domain that interacts with Cul2-Rbx1 to determine its binding specificity [5].
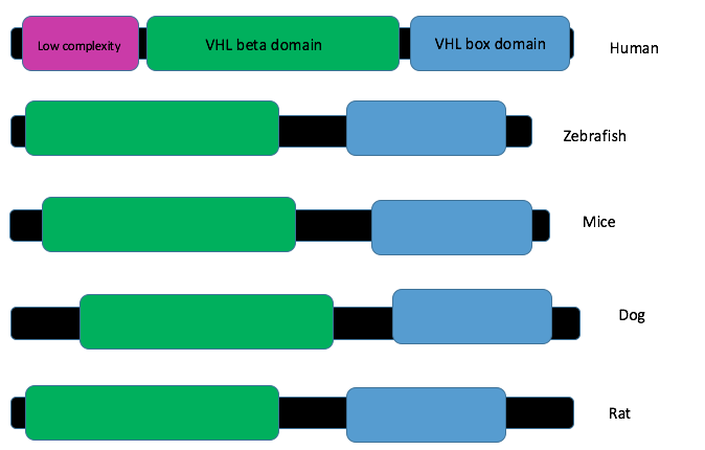
Are VHL protein domains present in other organsims? Yes!
Analysis
There is a high level of the VHL box and VHL beta domain across homologous organisms of VHL. However, homo sapiens are the only organism that contains a low complexity region. Because of this, I would like to further investigate the role of this low complexity region in the normal functioning of VHL.
References
1. EMBL-EBI: What are protein domains? <https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains>
2. Copley Richard R.,Doerks Tobias,Letunic Ivica and Bork Peer(2002), Protein domain analysis in the era of complete genomes, FEBS Letters, 513, doi: 10.1016/S0014-5793(01)03289-6
3. Coletta, A., Pinney, J. W., Solís, D. Y. W., Marsh, J., Pettifer, S. R., & Attwood, T. K. (2010). Low-complexity regions within protein sequences have position-dependent roles. BMC Systems Biology, 4, 43. http://doi.org/10.1186/1752-0509-4-43
4. EMBL:EBI: Summary: VHL beta domain. <http://pfam.xfam.org/family/PF01847.14>
5. Kamura, T., Maenaka, K., Kotoshiba, S., Matsumoto, M., Kohda, D., Conaway, R. C., … Nakayama, K. I. (2004). VHL-box and SOCS-box domains determine binding specificity for Cul2-Rbx1 and Cul5-Rbx2 modules of ubiquitin ligases. Genes & Development, 18(24), 3055–3065. http://doi.org/10.1101/gad.1252404
2. Copley Richard R.,Doerks Tobias,Letunic Ivica and Bork Peer(2002), Protein domain analysis in the era of complete genomes, FEBS Letters, 513, doi: 10.1016/S0014-5793(01)03289-6
3. Coletta, A., Pinney, J. W., Solís, D. Y. W., Marsh, J., Pettifer, S. R., & Attwood, T. K. (2010). Low-complexity regions within protein sequences have position-dependent roles. BMC Systems Biology, 4, 43. http://doi.org/10.1186/1752-0509-4-43
4. EMBL:EBI: Summary: VHL beta domain. <http://pfam.xfam.org/family/PF01847.14>
5. Kamura, T., Maenaka, K., Kotoshiba, S., Matsumoto, M., Kohda, D., Conaway, R. C., … Nakayama, K. I. (2004). VHL-box and SOCS-box domains determine binding specificity for Cul2-Rbx1 and Cul5-Rbx2 modules of ubiquitin ligases. Genes & Development, 18(24), 3055–3065. http://doi.org/10.1101/gad.1252404